Lead
Lead (Pb) is a silver-grey metal commonly found with zinc and copper sulphides as the mineral galena. Properties such as a low melting point (327.4°C), high density and malleability, corrosion resistance and a good ability to attenuate gamma radiation and sound vibration have led to a wide variety of uses. Lead oxide was used for pottery glazing as early as 7000 to 5000 BC, and lead was one of the earliest smelted metals. It was used for coinage in China and Greece, and about 100 BC the Romans began using it for plumbing and solder. Roofing and ammunition applications developed during the Middle Ages. Lead-acid batteries, which account for the largest use of lead today, were introduced in 1911. More recent applications capitalize on lead's ability to block gamma radiation and dampen sound vibration. Lead is used for electronic equipment, chemicals, alloys and cable sheathing, and as a protective barrier against nuclear radiation or X-rays from televisions, computer monitors and medical-dental equipment.
Lead is extracted from mined ores containing zinc, silver and minor amounts of copper. It is also recovered from recycled materials; more than 50% of world metal production comes from recycling, making lead one of the most recycled metals in the world. Canada mines approximately 149 000 tonnes of lead annually, refines about 285 000 tonnes and recycles about 126 000 tonnes.
Leading Provincial Producers
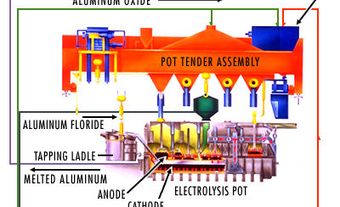
Leading Canadian producers are NB, BC, and Nunavut. Ore is mined, crushed and ground, and the lead-containing mineral is separated by flotation to produce a concentrate. At the smelter, concentrate is oxidized by heating to drive off sulphur, which is recovered as sulphuric acid or sulphur dioxide and may be further processed into fertilizers. The oxidized concentrate or recycled lead scrap is then smelted, usually in a blast furnace, to produce impure lead bullion. Electrolytic or thermal refining purifies the lead and recovers by-products, including silver, antimony and bismuth (see metallurgy). Primary smelters in BC and NB convert concentrates and recycled materials to refined metal and alloys. Secondary smelters in Québec, Ontario, Manitoba and BC also recycle spent products such as automobile batteries into metal and alloys.
Risks and Hazards
Lead is toxic at high levels of exposure and lead poisoning was formerly an occupational hazard (see pollution). Health-related issues with lead are often well documented and managed. Production and use of lead continues to rise while risks from exposure to lead have been greatly reduced. Average blood lead concentrations in Canadian children declined from about 19 micrograms per decilitre (µg/dL) in 1972 to 12 µg/dL in 1984 and 6µg/dL in 1988. Actions taken in Canada to reduce unsafe lead exposure range from regulations phasing lead out of gasoline to educational initiatives and voluntary industry action on lead in paint, lead-soldered food cans, occupational exposures and industrial emissions. These results demonstrate that lead can be used safely when properly managed.

 Share on Facebook
Share on Facebook Share on X
Share on X Share by Email
Share by Email Share on Google Classroom
Share on Google Classroom